[solved]-Create Phase Diagram Mixture Developing Curves Given Data Model Fusion Curve Power Equatio Q39030211
Create the phase diagram for the mixture by developing curveswith the given data. Model the fusion curve as a power equation andmodel the vaporization curve with an exponential curve using thedata from Boundaries.csv. Where these two curves intersect will bethe triple point of the mixture. Model the sublimation curve as alinear equation using the origin of the phase diagram (T = 150, P =0) and the triple point. On the phase diagram, the fusion curveshould begin at the triple point and end at the phase diagram upperlimit. The vaporization curve should begin at the triple point andend at the critical temperature. The sublimation curve should beginat T = 150, P = 0 and end at the triple point. An additionalboundary between the liquid and gas phases should appear as avertical line at the critical temperature.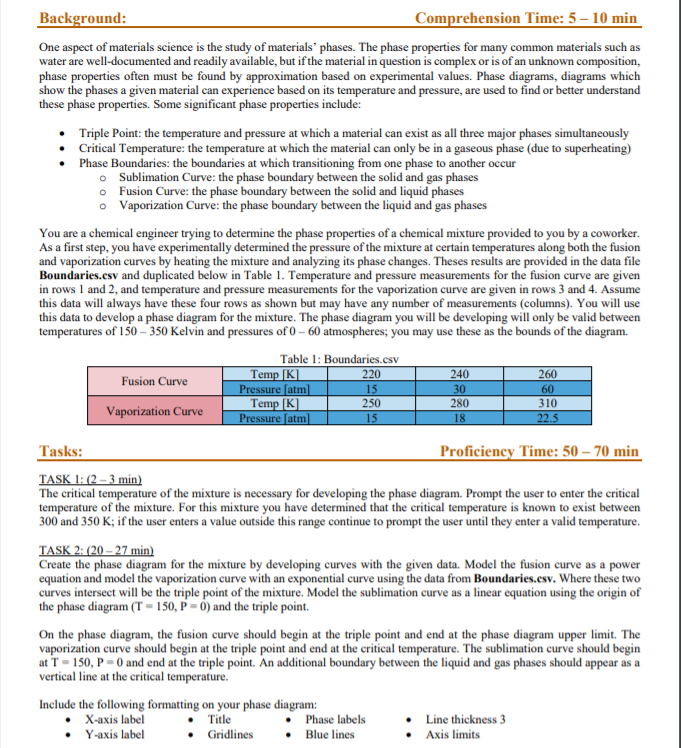

![Phase Diagram of Unknown Mixture Solid Liquid Pressure (P) (atm) Gas 150 200 250 300 350 Temperature (T) [K] Extra Credit Pha](https://media.cheggcdn.com/media/a92/s681x546/a926919e-4373-46e4-8836-6d3ee2fac5b8/phpc0Wmuq.png)
Background: Comprehension Time: 5-10 min One aspect of materials science is the study of materials’ phases. The phase properties for many common materials such as water are well-documented and readily available, but if the material in question is complex or is of an unknown composition, phase properties often must be found by approximation based on experimental values. Phase diagrams, diagrams which show the phases a given material can experience based on its temperature and pressure, are used to find or better understand these phase properties. Some significant phase properties include: • • Triple Point: the temperature and pressure at which a material can exist as all three major phases simultaneously Critical Temperature: the temperature at which the material can only be in a gaseous phase (due to superheating) Phase Boundaries: the boundaries at which transitioning from one phase to another occur Sublimation Curve: the phase boundary between the solid and gas phases Fusion Curve: the phase boundary between the solid and liquid phases Vaporization Curve: the phase boundary between the liquid and gas phases You are a chemical engineer trying to determine the phase properties of a chemical mixture provided to you by a coworker. As a first step, you have experimentally determined the pressure of the mixture at certain temperatures along both the fusion and vaporization curves by heating the mixture and analyzing its phase changes. Theses results are provided in the data file Boundaries.csv and duplicated below in Table 1. Temperature and pressure measurements for the fusion curve are given in rows 1 and 2, and temperature and pressure measurements for the vaporization curve are given in rows 3 and 4. Assume this data will always have these four rows as shown but may have any number of measurements (columns). You will use this data to develop a phase diagram for the mixture. The phase diagram you will be developing will only be valid between temperatures of 150 – 350 Kelvin and pressures of 0-60 atmospheres, you may use these as the bounds of the diagram. Fusion Curve Table 1: Boundaries.csv Temp [K] 220 Pressure ſatm 1 5 Temp K 250 Pressure (atm) 15 240 30 280 18 260 60 310 22.5 Vaporization Curve Tasks: Proficiency Time: 50 – 70 min TASK 1: (2-3 min) The critical temperature of the mixture is necessary for developing the phase diagram. Prompt the user to enter the critical temperature of the mixture. For this mixture you have determined that the critical temperature is known to exist between 300 and 350 K; if the user enters a value outside this range continue to prompt the user until they enter a valid temperature. TASK 2: (20 – 27 min) Create the phase diagram for the mixture by developing curves with the given data. Model the fusion curve as a power equation and model the vaporization curve with an exponential curve using the data from Boundaries.esy. Where these two curves intersect will be the triple point of the mixture. Model the sublimation curve as a linear equation using the origin of the phase diagram (T = 150, P=0) and the triple point. On the phase diagram, the fusion curve should begin at the triple point and end at the phase diagram upper limit. The vaporization curve should begin at the triple point and end at the critical temperature. The sublimation curve should begin at T = 150, P = 0 and end at the triple point. An additional boundary between the liquid and gas phases should appear as a vertical line at the critical temperature. Include the following formatting on your phase diagram: • X-axis label • Title Phase labels • Y-axis label • Gridlines . Blue lines • • Line thickness 3 Axis limits TASK 3: (18-24 min) With the phase diagram created, prompt the user to input a point to analyze on the phase diagram. Check that this point is within the phase diagram limits; if not, continue prompting the user until a valid point is entered. Create the function CheckPhase_cougarnetID.m that checks the phase of the user-inputted point. If the point is exactly on a phase boundary, you may choose which phase the point is classified as. The function should output a string array containing the phase of the point and should add that point to the phase diagram. Call your new function to determine the phase and output to the command window the phase of the entered point. TASK 4: (2-3 min) Ask the user if they would like to enter another point on the phase diagram. If yes, repeat Task 3. If no, continue to Task 5. TASK 5: (5-8 min) Output to the command window a table of the points inputted by the user and their corresponding phases. This table should include headers stating what each column represents. See sample output. For Task 5, you cannot use cell2table() or equivalent functions. TASK 6: (3-5 min) Find the percentage of the phase diagram that is in the gas phase. Output the percentage of the diagram in the gas phase to the command window. Extra Credit: -Number the points added to the phase diagram. -Create the text file Phase_results.txt which contains the table made in Task 5. Sample Output Command Window Enter the critical temperature of the mixture (K): 275 Enter the critical temperature of the mixture (W): 351 Enter the critical temperature of the mixture (X): 330 Enter a test point on the phase diagram (T, PJ: 1200 201 The point (200, 20) is in the solid phase. Enter a test point on the phase diagram (T, P): (300 301 The point (300, 30) is in the liquid phase. Enter a test point on the phase diagram (T, P: (300-10) Enter a test point on the phase diagram [T, P] : [119 10) Enter a test point on the phase diagram (T, P]: [300 101 The point (300, 10) is in the gas phase. Enter a test point on the phase diagram [T, P] : [330 50) The point (330, 50) is in the gas phase. Enter a test point on the phase diagram [T, P] : [225 50 The point (225, 50) 18 in the solid phase. Temperature [K] Pressure (atm) State 200 20 solid 300 11 quid 300 gas 330 solid 225 The gas phase is 30.278 of the phase diagram. Phase Diagram of Unknown Mixture Solid Liquid Pressure (P) (atm) Gas 150 200 250 300 350 Temperature (T) [K] Extra Credit Phase results.txt: 1 Temperature [K] Pressure (atm) 200 300 300 330 225 State solid liquid gas gas solid 20 Show transcribed image text Background: Comprehension Time: 5-10 min One aspect of materials science is the study of materials’ phases. The phase properties for many common materials such as water are well-documented and readily available, but if the material in question is complex or is of an unknown composition, phase properties often must be found by approximation based on experimental values. Phase diagrams, diagrams which show the phases a given material can experience based on its temperature and pressure, are used to find or better understand these phase properties. Some significant phase properties include: • • Triple Point: the temperature and pressure at which a material can exist as all three major phases simultaneously Critical Temperature: the temperature at which the material can only be in a gaseous phase (due to superheating) Phase Boundaries: the boundaries at which transitioning from one phase to another occur Sublimation Curve: the phase boundary between the solid and gas phases Fusion Curve: the phase boundary between the solid and liquid phases Vaporization Curve: the phase boundary between the liquid and gas phases You are a chemical engineer trying to determine the phase properties of a chemical mixture provided to you by a coworker. As a first step, you have experimentally determined the pressure of the mixture at certain temperatures along both the fusion and vaporization curves by heating the mixture and analyzing its phase changes. Theses results are provided in the data file Boundaries.csv and duplicated below in Table 1. Temperature and pressure measurements for the fusion curve are given in rows 1 and 2, and temperature and pressure measurements for the vaporization curve are given in rows 3 and 4. Assume this data will always have these four rows as shown but may have any number of measurements (columns). You will use this data to develop a phase diagram for the mixture. The phase diagram you will be developing will only be valid between temperatures of 150 – 350 Kelvin and pressures of 0-60 atmospheres, you may use these as the bounds of the diagram. Fusion Curve Table 1: Boundaries.csv Temp [K] 220 Pressure ſatm 1 5 Temp K 250 Pressure (atm) 15 240 30 280 18 260 60 310 22.5 Vaporization Curve Tasks: Proficiency Time: 50 – 70 min TASK 1: (2-3 min) The critical temperature of the mixture is necessary for developing the phase diagram. Prompt the user to enter the critical temperature of the mixture. For this mixture you have determined that the critical temperature is known to exist between 300 and 350 K; if the user enters a value outside this range continue to prompt the user until they enter a valid temperature. TASK 2: (20 – 27 min) Create the phase diagram for the mixture by developing curves with the given data. Model the fusion curve as a power equation and model the vaporization curve with an exponential curve using the data from Boundaries.esy. Where these two curves intersect will be the triple point of the mixture. Model the sublimation curve as a linear equation using the origin of the phase diagram (T = 150, P=0) and the triple point. On the phase diagram, the fusion curve should begin at the triple point and end at the phase diagram upper limit. The vaporization curve should begin at the triple point and end at the critical temperature. The sublimation curve should begin at T = 150, P = 0 and end at the triple point. An additional boundary between the liquid and gas phases should appear as a vertical line at the critical temperature. Include the following formatting on your phase diagram: • X-axis label • Title Phase labels • Y-axis label • Gridlines . Blue lines • • Line thickness 3 Axis limits
TASK 3: (18-24 min) With the phase diagram created, prompt the user to input a point to analyze on the phase diagram. Check that this point is within the phase diagram limits; if not, continue prompting the user until a valid point is entered. Create the function CheckPhase_cougarnetID.m that checks the phase of the user-inputted point. If the point is exactly on a phase boundary, you may choose which phase the point is classified as. The function should output a string array containing the phase of the point and should add that point to the phase diagram. Call your new function to determine the phase and output to the command window the phase of the entered point. TASK 4: (2-3 min) Ask the user if they would like to enter another point on the phase diagram. If yes, repeat Task 3. If no, continue to Task 5. TASK 5: (5-8 min) Output to the command window a table of the points inputted by the user and their corresponding phases. This table should include headers stating what each column represents. See sample output. For Task 5, you cannot use cell2table() or equivalent functions. TASK 6: (3-5 min) Find the percentage of the phase diagram that is in the gas phase. Output the percentage of the diagram in the gas phase to the command window. Extra Credit: -Number the points added to the phase diagram. -Create the text file Phase_results.txt which contains the table made in Task 5. Sample Output Command Window Enter the critical temperature of the mixture (K): 275 Enter the critical temperature of the mixture (W): 351 Enter the critical temperature of the mixture (X): 330 Enter a test point on the phase diagram (T, PJ: 1200 201 The point (200, 20) is in the solid phase. Enter a test point on the phase diagram (T, P): (300 301 The point (300, 30) is in the liquid phase. Enter a test point on the phase diagram (T, P: (300-10) Enter a test point on the phase diagram [T, P] : [119 10) Enter a test point on the phase diagram (T, P]: [300 101 The point (300, 10) is in the gas phase. Enter a test point on the phase diagram [T, P] : [330 50) The point (330, 50) is in the gas phase. Enter a test point on the phase diagram [T, P] : [225 50 The point (225, 50) 18 in the solid phase. Temperature [K] Pressure (atm) State 200 20 solid 300 11 quid 300 gas 330 solid 225 The gas phase is 30.278 of the phase diagram.
Phase Diagram of Unknown Mixture Solid Liquid Pressure (P) (atm) Gas 150 200 250 300 350 Temperature (T) [K] Extra Credit Phase results.txt: 1 Temperature [K] Pressure (atm) 200 300 300 330 225 State solid liquid gas gas solid 20
Expert Answer
Answer to Create the phase diagram for the mixture by developing curves with the given data. Model the fusion curve as a power equ… . . .
OR

