[Solved]3 Liquid Boils Vapor Pressure Equals External Pressure Acting Surface Liquid Water Boils L Q37298063
**PLEASE USE MATLAB 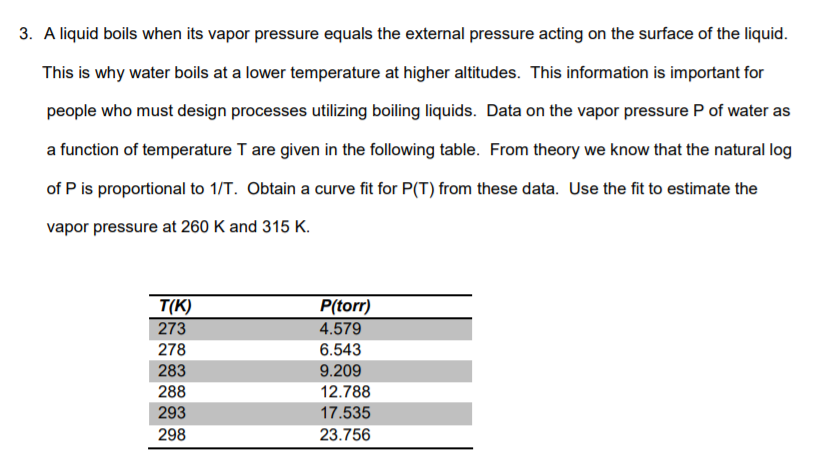
3. A liquid boils when its vapor pressure equals the external pressure acting on the surface of the liquid This is why water boils at a lower temperature at higher altitudes. This information is important for people who must design processes utilizing boiling liquids. Data on the vapor pressure P of water as a function of temperature T are given in the following table. From theory we know that the natural log of P is proportional to 1/T. Obtain a curve fit for P(T) from these data. Use the fit to estimate the vapor pressure at 260 K and 315 K. T(K) 273 278 283 288 293 298 P(torr) 4.579 6.543 9.209 12.788 17.535 23.756 Show transcribed image text 3. A liquid boils when its vapor pressure equals the external pressure acting on the surface of the liquid This is why water boils at a lower temperature at higher altitudes. This information is important for people who must design processes utilizing boiling liquids. Data on the vapor pressure P of water as a function of temperature T are given in the following table. From theory we know that the natural log of P is proportional to 1/T. Obtain a curve fit for P(T) from these data. Use the fit to estimate the vapor pressure at 260 K and 315 K. T(K) 273 278 283 288 293 298 P(torr) 4.579 6.543 9.209 12.788 17.535 23.756
Expert Answer
Answer to 3. A liquid boils when its vapor pressure equals the external pressure acting on the surface of the liquid This is why w… . . .
OR

